
For example, carbon has nine possible integer oxidation states from −4 to +4. Pollucite ores, found in Maine and South Dakota, USA contain about 13 Cs 2 O. Most elements have more than one possible oxidation state. The natural source yielding the greatest quantity of caesium is the rare mineral pollucite. Cesium, chemical element of Group 1 (also called Group Ia) of the periodic table, the alkali metal group, and the first element to be discovered spectroscopically (1860), by German scientists Robert Bunsen and Gustav Kirchhoff, who named it for the unique blue lines of its spectrum (from the Latin caesius, ‘sky-blue’). How many protons does cesium have, Oxidation states are typically represented by integers which may be positive, zero, or negative. How many electrons does Caesium have? NameĬaesium – Element information, properties and uses | Periodic Table. 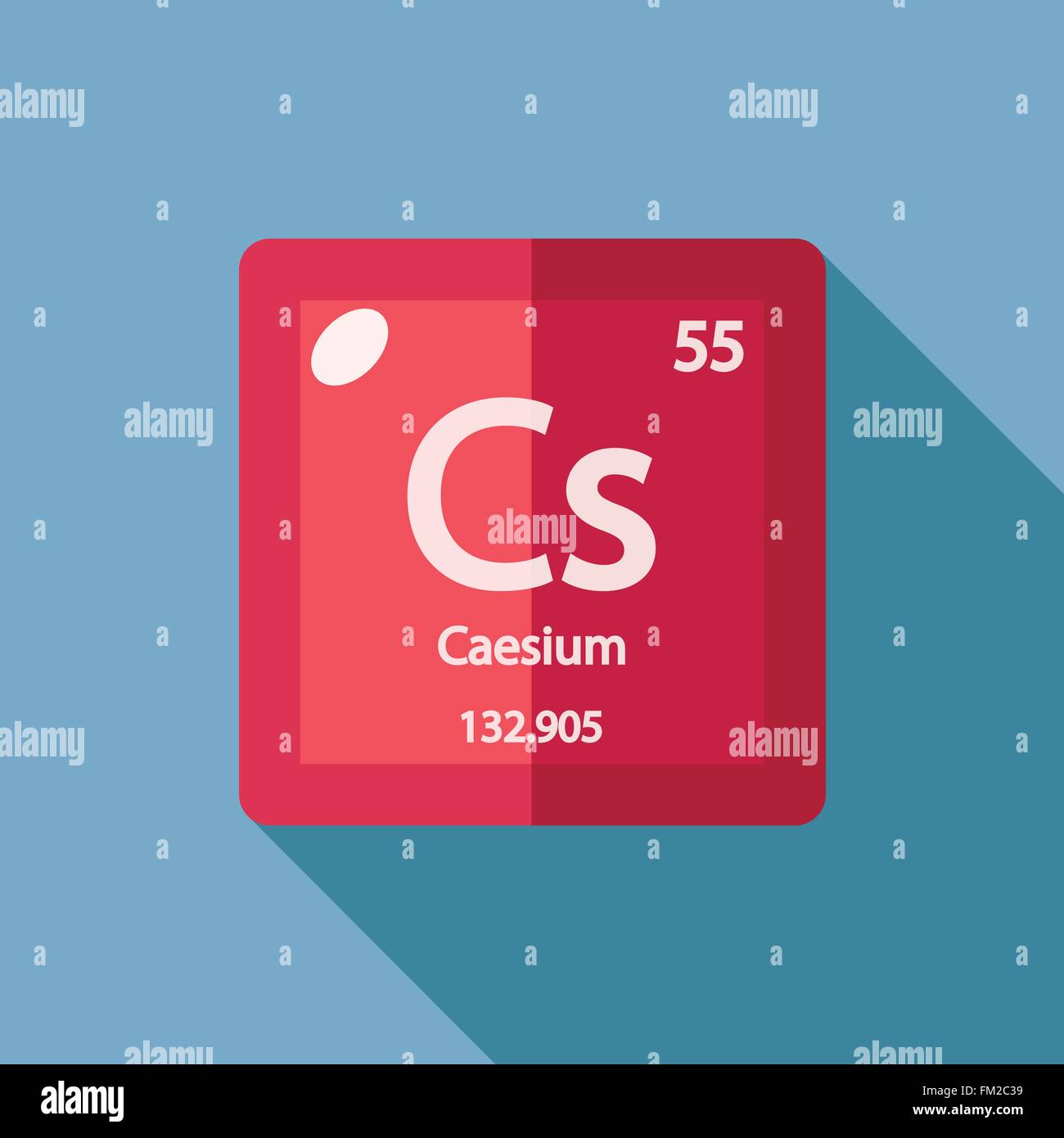
All the electrons but the outermost one are confined to orbits in stable shells. It so happens, the United States Naval Observatory explains, that cesium 133 atoms have their 55 electrons distributed in an ideal manner for this purpose. How many electrons are in an atom of cesium 133? So that means an atom of Cesium has 55 protons and if it’s an atom it’s neutral. An atom of Caesium has only 1 electron in its outer shell. How many protons neutrons and electrons are in cesium 133? NameĬaesium as 55 protons and 78 neutrons in its nucleus giving it an Atomic Number of 55 and an atomic mass of 133. How many protons neutrons and electrons does cesium have? Name The atomic number of each element increases by one, reading from left to right. Period A horizontal row in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. How many electrons are in an atom of cesium 133? Caesium 55 132.905 Glossary Group A vertical column in the periodic table.How many protons neutrons and electrons are in cesium 133?.How many protons neutrons and electrons does cesium have?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
